Tuesday Poster Session
Category: Colon
P4566 - GLP-1 Receptor Agonists Worsen Colonoscopy Quality Indicators: A Meta-Analysis of 18,805 Patients
.jpg)
Saqr Alsakarneh, MD, MS
Mayo Clinic
Rochester, MN
Presenting Author(s)
1Mayo Clinic, Kansas City, MO; 2Henry Ford Health, Detroit, MI; 3University of Missouri - Kansas City School of Medicine, Kansas City, MO; 4University of Kansas School of Medicine, Kansas City, KS; 5University of Colorado Anschutz Medical Campus, Denver, CO; 6NYU Grossman School of Medicine, Division of Gastroenterology and Hepatology, New York, NY
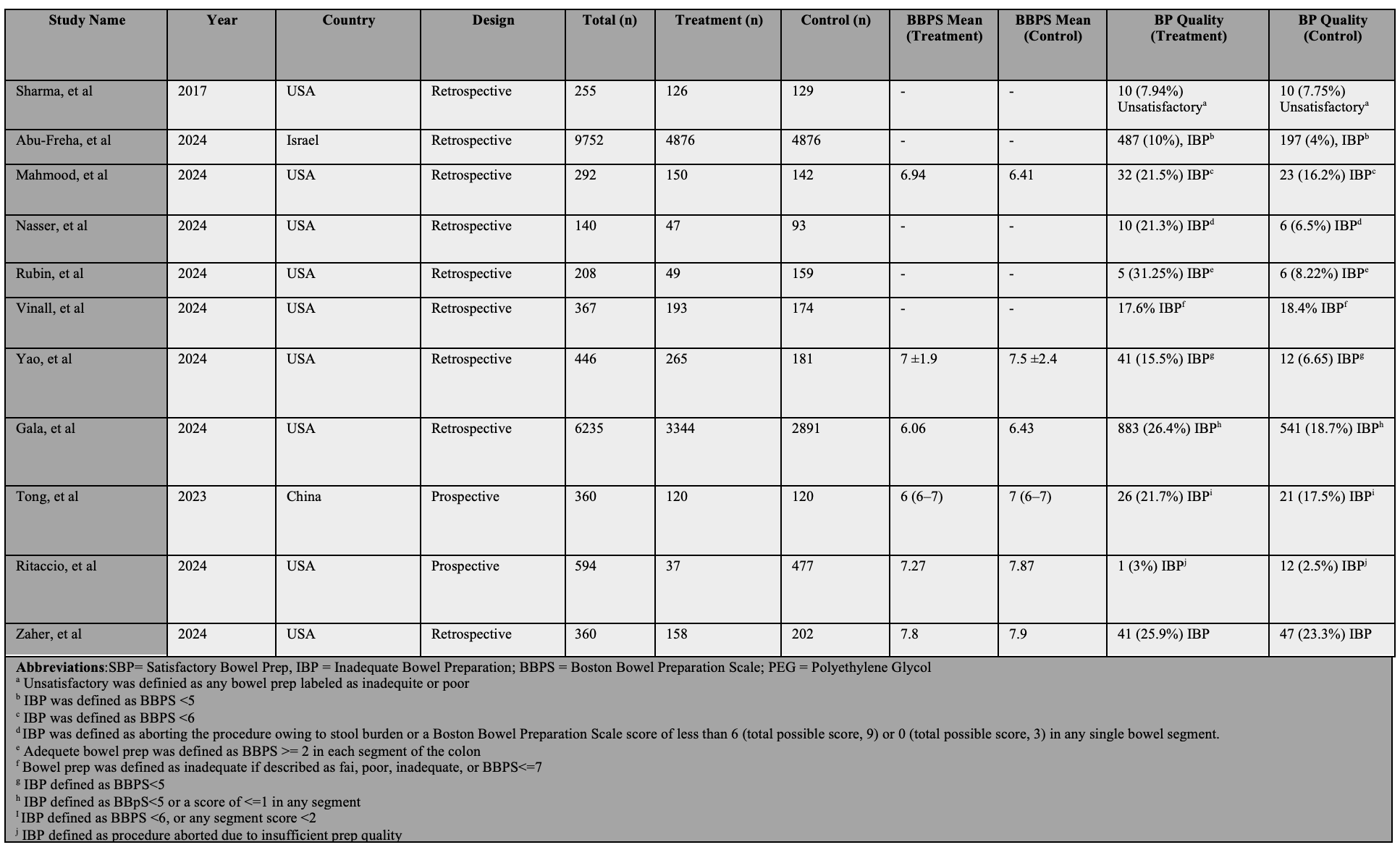
Introduction: Colonoscopy quality is critical for colorectal cancer screening and polyp detection. However, medications and comorbidities that slow gastrointestinal motility may negatively impact bowel preparation and hence the quality of colonoscopy. Previous reports suggested that Glucagon-like peptide-1 receptor agonists (GLP-1RAs) may impair bowel preparation due to their effects on gastrointestinal motility. This meta-analysis aims to evaluate the impact of GLP-1RAs on bowel preparation quality, including overall inadequate preparation (IBP) rates and differences in Boston Bowel Preparation Scale (BBPS) scores.
Methods: We conducted a comprehensive literature search of PubMed, Scopus, Embase, and Web of Science databases from inception to May 2025. We included studies that evaluated the effect of GLP-1RAs on bowel preparation quality. Pooled odds ratios (ORs) and mean differences (MDs) with 95% confidence intervals (CIs) were calculated using random-effects models and the Mantel-Haenszel method. Heterogeneity was assessed using the I² index. The primary outcome was IBP. Secondary outcomes included (1) BBPS scores, (2) repeat colonoscopy rates due to IBP, and (3) subgroup analysis among patients with diabetes.
Results:
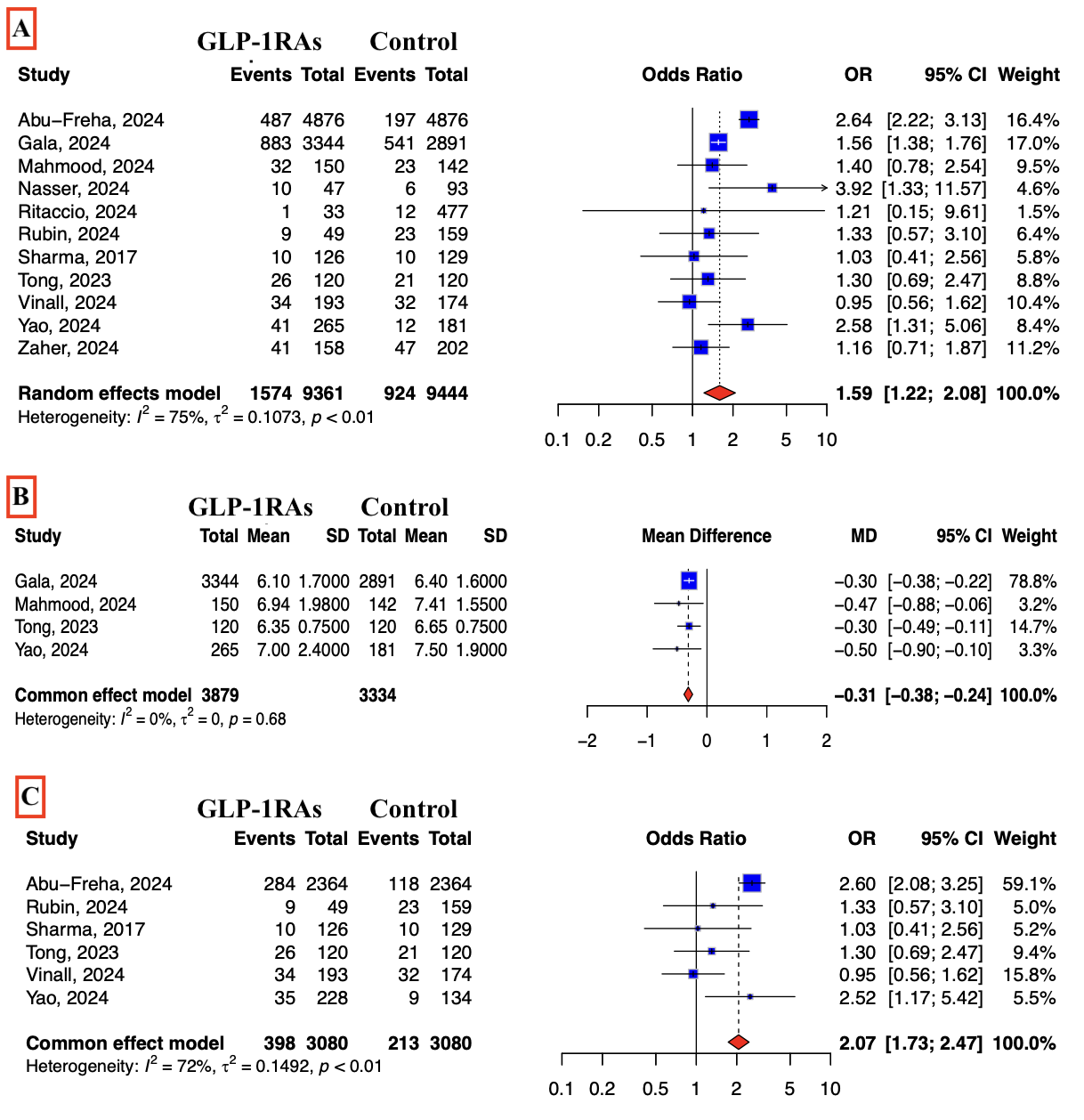
Our initial search resulted in 832 citations and 11 studies were included in the final analysis. A total of 18,805 patients were included (GLP-1RAs cohort, n=9,361; control, n=9,444) [Table 1]. GLP-1RAs use was associated with a significantly higher risk of IBP compared to the control group (16.8% vs 9.8%; OR = 1.59, [95% CI: 1.22 – 2.08], p < 0.01, Figure 1-A). Similarly, the mean BBPS score was lower in the GLP-1RAs group compared to the control group (MD = -0.31; 95% CI -0.38, -0.24; p < 0.001; Figure 1-B) with no observed heterogeneity between included studies (I2 = 0%). A sensitivity analysis among diabetic patients showed similar increased IBP risk (OR = 2.07, [95% CI: 1.73 – 2.47], p < 0.01, Figure 1-C). Additionally, GLP-1RA users had higher odds of requiring repeat colonoscopy due to IBP (OR = 1.77; 95% CI: 1.04–3.01; p=0.03).
Discussion:
GLP-1RAs are associated with a significantly increased risk of inadequate bowel preparation, lower BBPS scores, and higher repeat colonoscopy rates. These findings support considering temporary cessation of GLP-1RAs in patients undergoing colonoscopy, particularly those with additional risk factors for poor bowel prep.
Disclosures:
Saqr Alsakarneh, MD, MS1, Razan Aburumman, MD2, Mohamad Adam, MD3, Dushyant S. Dahiya, MD4, Mohammad Bilal, MD, FACG5, Aasma Shaukat, MD, MPH, FACG6. P4566 - GLP-1 Receptor Agonists Worsen Colonoscopy Quality Indicators: A Meta-Analysis of 18,805 Patients, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.
